Rapamycin (sirolimus) is one of the most promising anti-aging drugs around.
Various studies show rapamycin can extend lifespan (learn more here).
However, some people are concerned about potential negative side effects of rapamycin, such as immunosuppression (I explain why this likely isn’t that big of an issue here), and metabolic problems such as insulin resistance, the topic of this article.
An often-heard concern is that rapamycin can induce insulin resistance, and increase fatty acids (R,R,R,R).
Especially increased insulin resistance worries people, given increased insulin resistance is often a precursor to type 2 diabetes, and various aging-related diseases.
However, various experts believe that this increase in “insulin resistance” (and thus increased diabetes risk) is not the classic, dangerous form of insulin resistance.
And that this rapamycin-induced insulin resistance is mainly an expected consequence of what rapamycin does with the body (R,R).
Rapamycin-induced pseudo-diabetes?
Their argument is the following: high doses of rapamycin induce significant autophagy and bring cells in a sort of “fasting” state.
In such a fasting state, the body will start suppressing the uptake and processing of sugar, especially in non-brain tissues, in order to keep as much as possible glucose circulating in the blood so it can reach the brain (the brain mainly uses sugars to function).
This survival mechanism was important in prehistoric times when people often found little or no food for long periods. So the little sugar that circulated in their body would then mainly go to the brain. This way, the brain would still be fed, which is useful, given a working brain is needed to find food again as quickly as possible.
In order to achieve this, most non-brain tissues become “insulin resistant”. They process glucose much less, sparing most of the circulating sugar for the brain.
Rapamycin can induce such a fasting state, especially when taking the drug in higher doses and continuously. Rapamycin then causes peripheral, non-brain tissues to become “insulin resistant” so they take up less sugar.
When people who are taking rapamycin are in such a state, and then still consume normal meals with considerable amounts of sugars, this could lead to increased glucose levels, which some then wrongfully interpret as type 2 diabetes.
However, some scientists call this rapamycin-induced insulin resistance not real diabetes, but “pseudo-diabetes”, “starvation diabetes” or “hunger diabetes”.
In fact, various other interventions can lead to pseudo-diabetes, such as fasting, starvation, ketogenic diets (in which people mainly consume fats and little glucose), or antibodies that neutralize ACBP/DBI (an obesogenic protein that induces hunger) (R).
Already in 1846, Claude Bernard discovered increased levels of glucose in the urine of rabbits when they were fed carrots after a few days of fasting (I wonder how he found out about the increased levels of glucose in the urine) (R).
Despite the fact that such interventions can cause insulin resistance in the short-term, fasting, caloric restriction, and starvation can drastically improve, and in some cases even reverse, type 2 diabetes in the long-term (R,R,R,R).
In a somewhat similar way, the increase of fatty acids in the blood when people take rapamycin could also be caused due to a rapamycin-induced fasting state, in which fatty acids are mobilized to serve as energy sources for other tissues.
Insulin resistant but still healthy?
Furthermore, despite the fact that rapamycin sometimes induces insulin resistance and other metabolic “problems” in mice, their lifespan is not reduced, and in fact often increased.
One of the most famous studies showing that rapamycin caused insulin resistance in mice (R) still showed that rapamycin was associated with increased longevity.
In various studies in which rapamycin induced insulin resistance, the animals still were protected from negative consequences of insulin resistance, like diet-induced obesity (R) or kidney damage (R,R).
This is contrary to “classic” insulin resistance which actually shortens lifespan and increases the risk of diabetes, heart disease, and cancer.
So despite this pseudo-diabetes, lifespan and healthspan are often still extended.
Even more interestingly, various studies show that rapamycin can actually improve glucose metabolism and insulin resistance.
For example, in humans rapamycin improved glucose uptake and reduced insulin resistance when stimulating the mTOR pathway (by infusing people with a nutrient-rich mixture of amino acids) (R).
In rodents, rapamycin can improve glucose metabolism and insulin resistance in diabetic animals (R,R,R,R). Rapamycin can also reduce complications of diabetes, like kidney disease (nephropathy) (R,R,R,R).
In cancer studies in humans, there is a trend showing that rapamycin-induced insulin resistance is associated with better outcomes, though this was statistically not significant (R) (do note that the doses of rapamycin in cancer are often higher and continuous (daily) compared to doses for longevity, and that rapamycin is often associated with other drugs that also can induce insulin resistance).
Studies show that – as expected- this rapamycin-induced insulin resistance is reversible (R,R,R,R).
Rapamycin is an mTOR inhibitor. Doing the contrary, namely stimulating mTOR, is well-known to cause insulin resistance, diabetes, metabolic dysregulation, accelerated aging and shortened lifespan (R,R).
In other words, some scientists believe that insulin resistance due to activation of mTOR shortens lifespan, while insulin resistance due to inhibition of mTOR increases lifespan (R).
Does rapamycin always induce insulin resistance?
Rapamycin, especially when given continuously and in high doses can induce insulin resistance. However, when administering rapamycin not daily, but intermittently, this insulin resistance can be substantially reduced or prevented.
For example, in one study mice were given rapamycin only every 5 days, and rapamycin didn’t impair blood glucose levels or insulin tolerance (R):
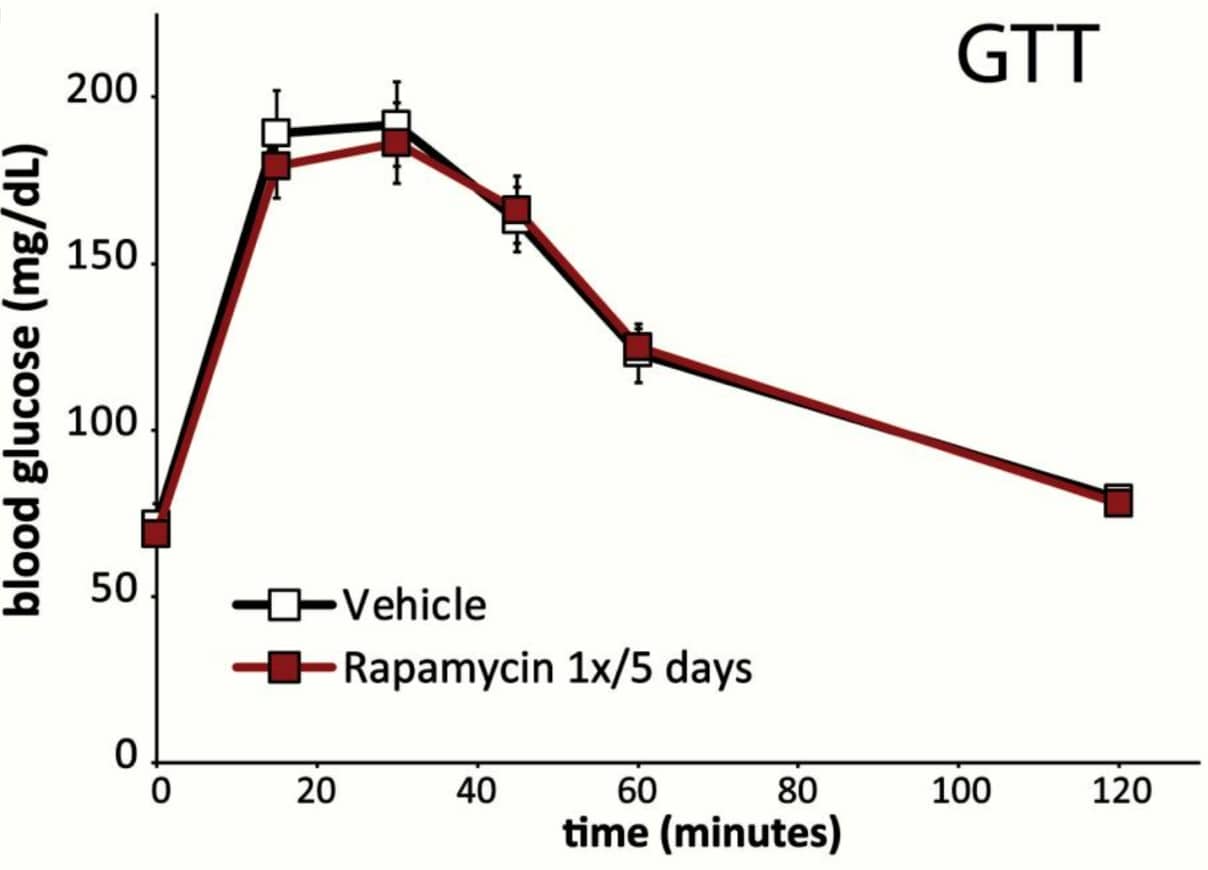
Blood glucose levels of mice treated with rapamycin are not increased (red line).
Interestingly, in this study, despite rapamycin was given intermittently, lifespan was increased by about 13 percent:
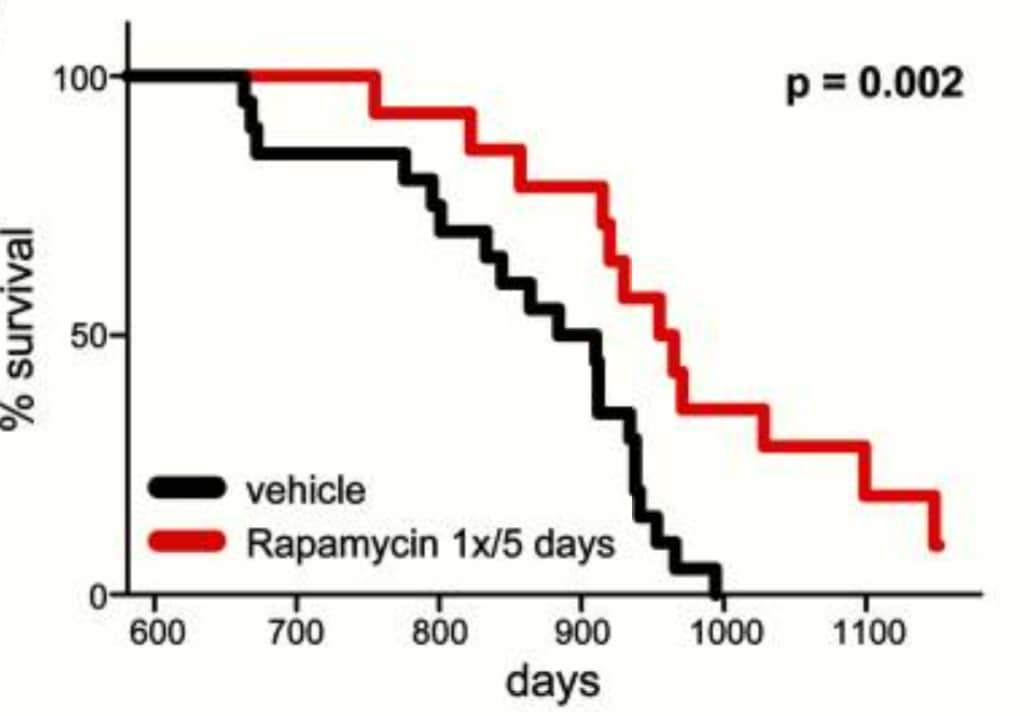
Lifespan is increased in mice receiving rapamycin (red line) versus controls (black line).
Even when given daily, rapamycin, when the dose is not too high, does not always lead to increased glucose levels or insulin resistance.
For example, in a study people received 1 mg of rapamycin daily. After 2 months there weren’t negative changes in their glucose metabolism (blood glucose concentration, insulin sensitivity and insulin secretion)(R).
So when given in a lower dose and/or not-continuously, the risk of rapamycin causing glucose intolerance or insulin resistance is significantly reduced.
One could even further reduce this risk by adding acarbose or metformin to rapamycin.
Metformin is a prescription drug that has been used for decades to treat type 2 diabetes. Metformin reduces insulin resistance, and also has been shown to extend lifespan (we discuss metformin for anti-aging later on). In diabetes-prone mice, adding metformin to rapamycin normalized insulin resistance (R).
Acarbose is probably even a better alternative to metformin. This drug stays in the gut and hinders the uptake of glucose into the blood (by inhibiting a protein in the gut that breaks down starches into glucose). Acarbose has been shown to extend lifespan, and works even better with rapamycin in this regard (R,R).
I’ll go deeper into acarbose later on.
Learn more about rapamycin:
- Does Rapamycin Cause High Glucose, Lipids and Diabetes?
- Does Rapamycin Increase Infection Risk?
- How Does Rapamycin Work? Overview of Rapamycin Lifespan Extension Studies
- Rapamycin for Anti-Aging and Longevity
- How does rapamycin work? How can it slow aging and extend lifespan?
Dr. Verburgh will publish in a few months his new book;
likely the best book on longevity for the general public!
To stay updated, subscribe to our popular longevity newsletter!