If you are male and an anti-aging doctor or guru recommends taking testosterone for longevity: run.
There are two main reasons why to run away: or this person is not up to date with insights into aging of the last 30 years, or they just want you to sell to you testosterone products and treatments.
It’s remarkable that many people still believe that increasing male hormones like testosterone is good for longevity.
Whether from an evolutionary, biological, and clinical perspective, male hormones like testosterone accelerate aging and shorten lifespan.
“Male hormones” include testosterone, DHEA (dehydroepiandrosterone) and dihydrotestosterone. For the main part, I will refer to testosterone as the prime example of a male hormone, but the things mentioned here can also account for other male hormones.
Before we explain how and why male hormones like testosterone accelerate aging, let us first clear some common misunderstandings which keep circulating and keep making people believe that testosterone is good for anti-aging.
1. “Testosterone makes me feel better”
Some men taking testosterone (or other male hormones) do feel better: they have more energy, more libido or more stamina. This can be due to the placebo effect (just thinking that a treatment could work can often make you feel better, even if it doesn’t work).
Nonetheless, there are studies showing that male hormones like testosterone can in some men indeed increase energy levels, mood or libido (R). But that doesn’t necessarily mean this drug is good for you in the long term.
2. Most studies with testosterone are short-term
Studies “demonstrating” that testosterone is healthy are often short-term studies, while looking at specific (crude) biomarkers of health.
For example, extra testosterone could reduce belly fat or increase muscle mass in some men. But these are short-term effects; perhaps in the long-term testosterone could increase heart disease risk. Or mortality.
Second, reduced belly fat or increased muscle mass are just small parts of the complete picture. Even if testosterone reduces belly fat or improves some (crude) metabolic biomarkers, this doesn’t necessarily mean it’s healthy. Perhaps testosterone can have many other, negative effects on the body which have not been measured in this study.
3. “Men with low testosterone have increased mortality”
There are studies showing that people with low testosterone have a shortened lifespan or are more unhealthy.
But this is likely an association, not a causative effect. People who are sick or unhealthy often have low testosterone levels because testosterone levels are reduced due to sickness, starvation, weight loss or an unhealthy lifestyle in general.
These people also have an increased risk of dying, because they are unhealthy, sick or lost weight due to (unknowingly) having cancer, HIV, or other diseases. In this way, low testosterone and shortened lifespan are associated, but this doesn’t necessarily mean that low testosterone causes increased morbidity (sickness) and mortality.
For example, people who are obese and/or have type 2 diabetes, often have lower levels of testosterone. This doesn’t imply that low testosterone causes diabetes. In fact, giving these patients extra testosterone (as has been done in some studies) is also not advisable. Firstly, the main cause of this testosterone deficiency needs to be addressed, which is an unhealthy diet. Secondly, you don’t want to give these patients another substance (testosterone) that accelerates aging on top of insulin, which also accelerates aging.
If changing the diet is really not an option (which should never be the case), then there are better alternatives than testosterone, as I discuss in the chapter about treating (and reversing) type 2 diabetes.
4. Real testosterone deficiency is rare
It’s often bandied about that testosterone levels go down precipitously during aging, leading to significant “testosterone deficiency”, also called “low T”.
But most elderly men are not testosterone deficient.
Real testosterone deficiency happens when people have serious health issues that significantly reduce testosterone levels, such as genetic mutations or damage to the pituitary gland which regulates testosterone production. Or they have damage to the gonads which produce testosterone. In these cases, of course, testosterone can be useful.
However, declining testosterone levels during aging are not serious testosterone deficiencies in most cases.
In fact, it could be that somewhat lower testosterone levels during aging is actually a good thing, as we will discuss further down.
But do testosterone levels actually really decline a lot during aging? There is still debate about this.
There are indeed studies showing that testosterone levels decline significantly during aging. But there are also other studies that show only a very mild decline.
For example, a study found that testosterone levels decline only a little bit during aging, from around 15.4 nmol/liter of blood at age 19, to 13 nmol/L at age 40, with no further fall after that (R) (After age 40, there can be more outliers (variance), which means there are old people who have too little or too much testosterone. However, these outliers also often already had much lower or higher cholesterol levels when they were young.)
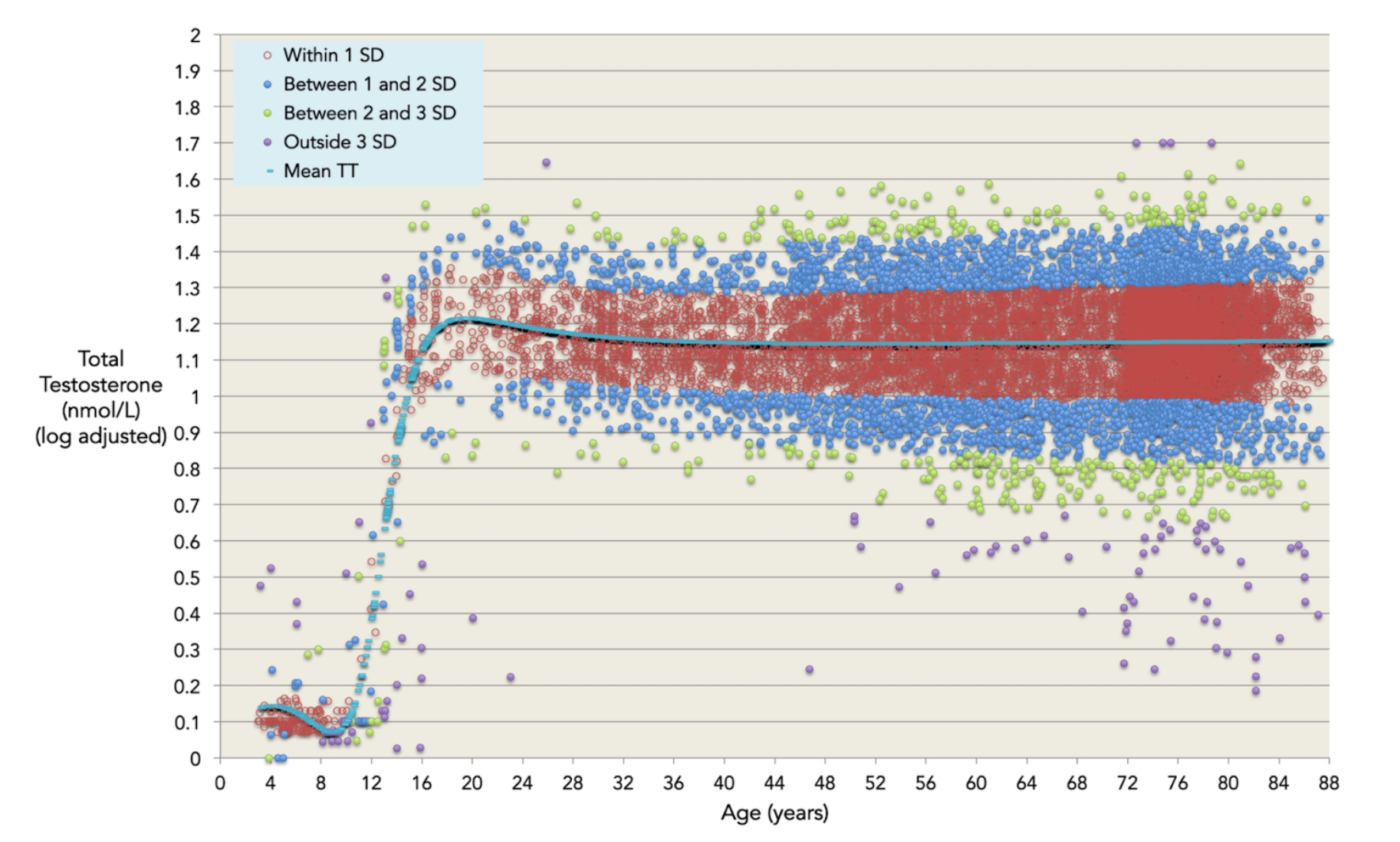
But as mentioned before, some studies do find that testosterone levels decline during aging. However, this could also be explained by the fact that with age many people become more unhealthy. For example, they become overweight, diabetic or get heart disease. These diseases and afflictions often lead to less testosterone production. Some scientists found that testosterone levels hardly decline in healthy old men (R).
Additionally, many testosterone studies use somewhat arbitrary, absolute cut-off values, e.g. “people are testosterone deficient if they have less than 11.3 nmol/L of testosterone”. But these are absolute values, not relative values. There can be old people with low testosterone values according to this definition, but some of them already had low testosterone levels from a young age on, making that during aging their testosterone levels hardly declined.
Also, what is a “low testosterone level”? It could be that people who have “low testosterone” according to one cut-of value, have no health problems, and do and feel great.
And even if people do have too low testosterone levels, this is not necessarily the reason why they feel tired or crappy or have low libido. There can be many other reasons for this, like not consuming enough B vitamins, omega-3 fatty acids and zinc for proper brain function so you feel more depressed, suffering from magnesium and iron deficiency so you feel more tired, having increased inflammaging due to aging, or cardiovascular disease leading to less blood flow to muscles and the penis (leading to fatigue and reduced erections), having an altered gut microbiome, and thousands of other reasons and mechanisms besides “low T”.
5. “It’s giving what is missing, not giving extra testosterone”
Often, testosterone proponents say that it’s not about giving extra testosterone, it’s just about adding some testosterone for men who have too little testosterone. So adding what is missing; bringing testosterone levels “back to normal”.
Firstly, studies found that in elderly people, the risk of heart disease was about 60% higher with testosterone therapy (R). These are mainly old males, of which many – according to testosterone-proponents – would need extra testosterone given “testosterone levels decline with age anyhow in most people”.
There are also studies showing that giving extra testosterone to people with low testosterone still increases their risk of heart attacks, stroke and mortality (R,R,R). So this popular we-just-add-what-is-missing testosterone mantra doesn’t fly.
Testosterone and (shortened) lifespan
Now we have cleared some of the most common testosterone misconceptions, let us look into the science about male hormones and lifespan and healthspan.
First, many studies in animals show that if you give male animals extra male hormones, like testosterone, they die faster (R).
DHEA, a more “mild androgen” and a precursor to male hormones like testosterone, could also shorten lifespan. In a study in which mice were given DHEA, these mice lived 25% shorter compared to the control group that didn’t receive DHEA (R).
The opposite is also true: if you castrate male organisms, they tend to live longer. Castrating male salmon can double their lifespan (R). Removing the gonads of C elegans worms increases their lifespan by more than 60%. Combining castration with inactivation of pro-growth pathways like the insulin/insulin-like-growth factor pathway can further increase their lifespan up to sixfold (R).
This seems also to be the case for humans: male eunuchs live longer, as do other castrated men (in earlier times, some mentally retarded men were castrated, a practice that wouldn’t be condoned in current times). It’s estimated that castrated men live on average 14 years longer (R,R,R).
It’s important to note that castration involves removing the testicles, so that very little testosterone is produced in the body. Some people think that “castration” also includes a modern surgical method to prevent men getting kids, but this is something else: it’s a vasectomy, in which the small vessel between the testes and penis are cut, so no sperm is produced during orgasm. Men cannot ejaculate anymore, but they still have testes that produce testosterone.
Also, having less sex won’t really make you live longer, given one still has testes that produce testosterone.
It’s also known that androgens (male hormones) suppress the immune system (R,R). Actually, reducing androgen levels could be one way to slow down the aging-related shrinking of the thymus, which is a gland behind our chest bone that produces immune cells. This could also help to explain why men are more vulnerable to infections compared to women (R,R) (their immune system is less strong), but also why women could be more prone to auto-immune diseases (given they have less androgens so they have a stronger, more active immune system).
Testosterone replacement therapy and disease and mortality risk in humans
Various studies show that giving men extra testosterone increases risk of various aging-related diseases and increases the risk of dying (R,R,R,R).
One study followed more than 15,000 men with low testosterone and found that people who received testosterone replacement therapy had up to 44% more risk of heart attacks and strokes (R). According to the researchers: “In the absence of identifiable causes of hypogonadism [like damage of dysfunction of the testis or pituitary gland], testosterone replacement therapy should be initiated with caution among aging men with low testosterone levels.”
In another study, elderly men with low testosterone who received testosterone were several times more likely to get a heart attack versus the placebo group (R). This study was ended prematurely, because it was deemed unethical to keep giving the elderly men testosterone given that the testosterone group had significantly more risk of a heart attack.
However, the men who received testosterone performed considerably better on leg-press and chest-press tests. So you can perform better at the gym, but you run a higher risk of getting a heart attack.
A meta-analysis ( a study that looks at hundreds of other studies) found that testosterone therapy increases the risk of a cardiovascular event (a heart attack or stroke) by 54%.
Interestingly, this meta-analysis also found that the effect of testosterone therapy varied with the source of funding. In other words, if the studies were not funded by pharmaceutical companies that bring testosterone to market, the risk of a heart attack was even bigger: 200% (a two fold increase).
However, when the study was funded by the pharmaceutical industry, the risk of a cardiovascular event was actually 11% lower with testosterone therapy (R). This brings us to the next point:
Pro-testosterone replacement therapy bias
The sexual health market is a very big one and there is lots of money at stake. This leads to many badly conducted studies, citation biases, and other kinds of dubious “scientific evidence” supporting testosterone replacement therapy.
In other words, be (also) critical of studies demonstrating that testosterone therapy has health effects; first check if they are sponsored by the pharmaceutical industry.
Another problem with lots of testosterone studies is that they are short-term, some even as short as three months. So of course, most people won’t get a heart attack after just three months. But what if you take extra testosterone for years? Or what happens after decades?
Another confounding factor is that often more wealthy and health-minded people have access to doctors that prescribe testosterone therapy. Often these people engage in many healthy behaviors that could partially offset the increased risk of heart disease and mortality with testosterone therapy.
Anyhow, the FDA required companies that sell testosterone to put a cardiovascular risk warning on all their testosterone products. And the FDA also restricted the use of testosterone only to people with hypogonadism. These are patients who have serious testosterone deficiency caused by serious problems, like damage or disease of the pituitary gland and testicular glands.
The FDA recommends testosterone replacement therapy only for these health indications, not for the common, aging-related decline in testosterone levels. In other words, not for “anti-aging” purposes, for which most testosterone products are marketed currently.
Why testosterone accelerates aging

Biologically speaking, it makes a lot of sense why more sex hormones like testosterone accelerate aging.
First, there is the “disposable soma theory”, which states that sex comes at the expense of longevity.
In nature, there is a tradeoff between sex and longevity. There is only so much energy available for an organism to maintain itself (and thus to slow aging). When it has to spend energy for reproduction this is at the expense of longevity.
In other words, energy spent on reproduction cannot be spent on maintaining the body to better withstand the ravages of time.
We see examples of this mechanism everywhere in nature. Mice have a very short lifespan but they reproduce a lot. Mice have big litters (eight to ten little mice) and can reproduce five to ten times a year. Salmon only reproduce once, but die quickly after that, given the sex hormones and cortisol (which make them fertile and ready to engage in their once-in-a-lifetime mating onslaught) quickly age them, damaging their bodies beyond repair.
Bowhead whales on the other hand give birth to just one calf every three to seven years. Humans, also a very long-lived species, reproduce only once or a few times over their entire lifespan.
So everywhere in nature, we see that more and faster reproduction (and increased sex hormones) is associated with faster aging and a shorter lifespan.
This could also help to explain why aging really starts to get going after puberty; at least according to some scientists. They claim we begin to really age when puberty starts.
A young child has a very low mortality risk. For example, an 11-year old has a mortality risk of about 1 in 40,000 in western countries. If his mortality risk would stay constant over his lifespan, this child could become on average 1,200 years old.
But this is of course not the case. The mortality risk starts to drastically increase (starting to double each 8 years) when puberty starts.
With the onset of puberty, the body becomes engulfed in sex hormones, which lead to sexual characteristics such as more muscle mass, body hair, breasts, beard growth, a deeper voice. However, with this new development phase, strongly induced and regulated by sex hormones, the body starts its accelerated pathway to old age and death.
We also see in nature that the age at when puberty kicks in (when organisms become fertile due to an increase in sex hormones) is strongly associated with total lifespan.
One way biologists estimate the lifespan of animals is by multiplying by six the age at which they sexually mature. So in humans, puberty starts at around the age of 13, and our average lifespan is around 78 years (13 x 6). Long-lived Aldabra turtles take about 25 years to sexually mature, and their lifespan is likely at least 150 years (25 x 6). This again shows the strong relation between reproduction (and reproductive sex hormones) and the rate of aging and lifespan.
Then there is also the antagonistic pleiotropy theory: what can be good for you when you are young can later on be bad for you. For example, having more testosterone in one’s teens and twenties can provide one with more muscle mass, strength, and libido, but might accelerate aging in the long term. One can also see this with your own eyes: there are men that look “very manly”: they have lots of muscle mass, a deep voice, pronounced cheekbones, and are big and strong. But they for example go bald early, and look like they are ten years older than their age. It could be that their sex-hormone-driven developmental program runs faster.
Are all sex hormones bad for longevity?
Does this mean that all sex hormones are detrimental and accelerate aging? Probably yes in the case of male sex hormones like testosterone.
However, with female sex hormones it’s more complicated.
Women live on average 6 years longer than men, and one reason for this is that they have less testosterone and more estrogen in their body.
A specific estrogen, namely 17-beta estradiol can extend lifespan. When male mice are given this female hormone they live longer (R,R). It doesn’t seem to work as well in castrated male mice, which makes sense given they already have less testosterone so adding estrogen will have less of an effect (R).
Gonad swapping also extends lifespan: if you give male mice ovaries they live longer (R). And removing the ovaries in women before menopause is associated with increased risk of aging diseases and reduced lifespan, an effect that was somewhat lessened when they received estrogen therapy (R,R,R,R).
In other studies, the ovaries were removed in mice before puberty, but at old age, these mice received young transplanted ovaries again. This increased their lifespan by 40% (R). Just giving old mice young ovaries also extended their lifespan (R).
So it could be that female reproductive organs, and associated female reproductive hormones like estrogen, could be beneficial for longevity. It could make sense evolutionary speaking, given women are more important and valuable for nature given they bring forth and help grow up children.
These insights could be interesting with regards to hormone replacement therapy (HRT) during menopause. I will discuss this later on. It’s a complicated topic in which the devil is in the details.